CONDENSATION SYNTHESIS
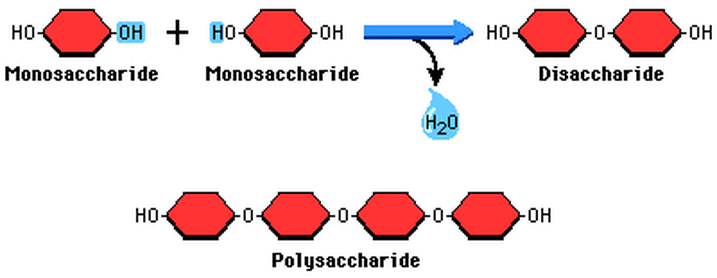
Condensation synthesis is the process by which two molecules are combined to make a dimer. Condensation synthesis begins with the removal of an -H and an -OH group from the molecule. This joins the two molecules together forming a dimer. More molecules can be added onto the dimer through condensation synthesis which leads to the production of complex biological molecules. The -H and -OH groups removed during condensation synthesis combine to form a water molecule.
Condensation synthesis is the process by which two molecules are combined to make a dimer. Condensation synthesis begins with the removal of an -H and an -OH group from the molecule. This joins the two molecules together forming a dimer. More molecules can be added onto the dimer through condensation synthesis which leads to the production of complex biological molecules. The -H and -OH groups removed during condensation synthesis combine to form a water molecule.
Bonds formed through condensation synthesis:
- glycosidic linkages (between two monosaccharides)
- ester bonds (between a glycerol and a fatty acid)
- peptide bonds (between two amino acids)
HYDROLYSIS
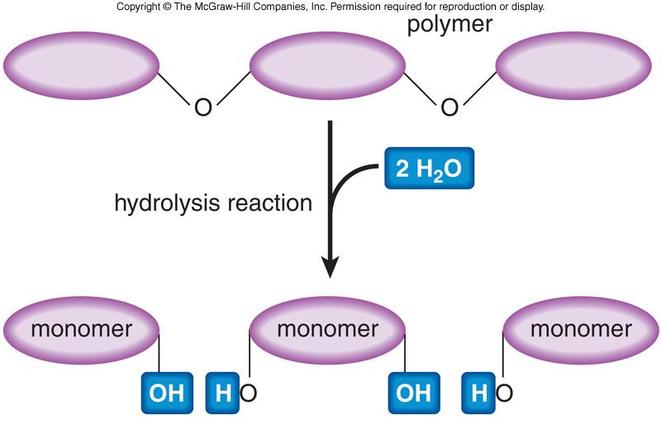
Hydrolysis is the process by which dimers, trimers, and polymers are broken down into monomers. Hydrolysis begins with the splitting of a water molecule. The water molecule splits into an -H group and an -OH. The -H and -OH groups are added to a macromolecule which breaks down the macromolecule into simple monomers.
Hydrolysis is the process by which dimers, trimers, and polymers are broken down into monomers. Hydrolysis begins with the splitting of a water molecule. The water molecule splits into an -H group and an -OH. The -H and -OH groups are added to a macromolecule which breaks down the macromolecule into simple monomers.