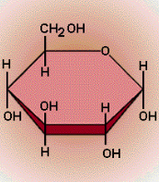
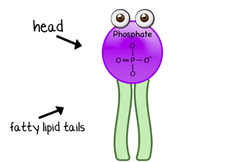
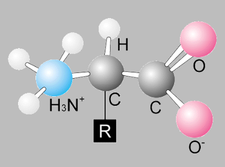
- Building Blocks- Monosaccharides
- Carbonyl and Hydroxyl Group causes the molecule to be polar
- Hydrophilic in water
Fats
- Building Blocks- Glycerol and Hydrocarbon chain
- Three carbon alcohol with fatty acid chain causes the molecule to be non-polar
- Hydrophobic in water
- Building Blocks- Glycerol, hydrocarbon chain, and phosphate group
- Phosphate group makes head polar and the tail is non-polar
- Amphipathic in water
- Building Blocks- hydrocarbons and carbon rings
- hydrocarbons and methyl group causes the molecule to be non-polar
- Hydrophobic in water
- Building Blocks- 4 fused C-rings
- Methyl group causes molecule to be non-polar but it has one hydroxyl group causing some polarity.
- Amphipathic in water
- Building Blocks- amino acids
- Amino group, Carboxyl group and a side chain that determines the polarity
- Depending on the functional group the side chain is, it can either be hydrophobic or hydrophilic in water
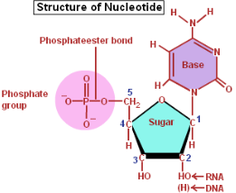
- Building Blocks- Nucleotides or Deoxynucleotides
- Ribose or deoxyribose and phosphate group cause the molecule to be polar
- Hydrophilic in water